From technology to system architecture: supporting the introduction of Pluslife near point-of-care TB diagnostics
Faster and more accurate tuberculosis (TB) diagnosis can mean earlier treatment initiation, reduced transmission, and stronger national response capacity. In high-burden settings where access to diagnostics remains limited, delays in diagnosis can also contribute to loss to follow-up and missed opportunities for active case finding. These delays are not only clinical challenges — they are system challenges.
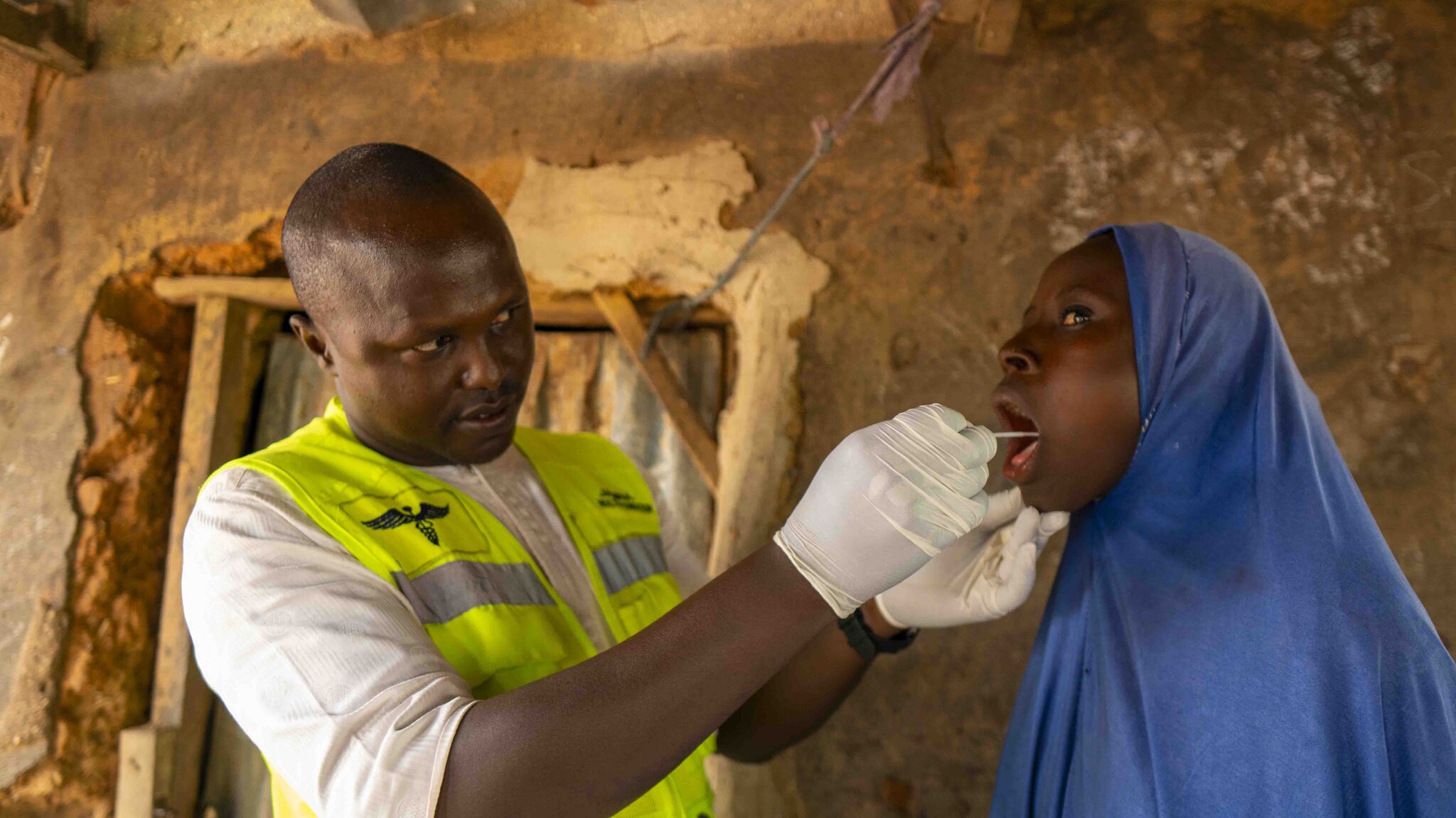
Near point-of-care molecular platforms such as the Pluslife MiniDock Ultra offer the potential to bring high-quality TB testing closer to primary care settings and patients, shortening the time between patient presentation and confirmed diagnosis. Recent technical guidance supporting decentralized molecular testing — including tongue swab sampling — signals a growing shift in the global TB response toward more accessible diagnostic models.
Across 13 countries, PFSCM is supporting preparatory efforts for the introduction of the Pluslife MiniDock Ultra, a near point-of-care molecular TB diagnostic device designed for decentralized use. This is not a routine product rollout. It represents a structured, early-stage multistakeholder effort to enable multi-country implementation of innovative TB technology.
Why this innovation matters
TB continues to disproportionately affect communities where centralized laboratory access is limited and diagnostic delays remain a barrier to timely care.
The Pluslife platform delivers molecular TB results in about 30 minutes and is engineered to operate in decentralized and resource-constrained environments, including high-temperature and high-humidity conditions. It supports both sputum and tongue swab sampling, offering greater flexibility in patient testing pathways.
Less invasive sample collection methods may also help expand access to testing for populations that are often harder to diagnose, particularly children and other patients who are unable to produce sputum.
For many low to middle-income countries, this shift is significant.
Diagnostics that can function closer to primary care settings help reduce structural access barriers, strengthen case detection, and support faster clinical decision-making. By narrowing the gap between patient presentation and confirmed diagnosis, near point-of-care testing has the potential to make national TB programs more responsive and resilient.
Innovation matters most when it moves closer to communities.
From technology to system architecture
Introducing a new class of TB diagnostics across 13 countries requires far more than procurement.
It demands regulatory alignment, supplier onboarding into complex public-sector ecosystems, multi-country coordination, shipment engineering, and anticipatory risk management – all before product movement begins.
It also requires shaping how the technology will be operationalized in-country, including service delivery models, training approaches, and clarity on roles across the supply chain.
PFSCM's role in supporting the early implementation of the Pluslife platform has also been highlighted in recent updates from the manufacturer, reflecting the level of coordination required to introduce new TB diagnostic technologies across multiple countries.
To enable a stable, scalable rollout, PFSCM has:
- Acted as a coordinating partner with global stakeholders, surfacing potential operational misalignments early and enabling structured dialogue to support smooth implementation.
- Contributed to a coordinated, cross-functional execution model integrating sourcing, quality assurance, logistics, and client services across PFSCM and partner stakeholders.
- Supported regulatory positioning across markets to reduce the risk of importation delays and inconsistent registration.
- Worked with Pluslife to clarify and strengthen the service delivery model for the platform, including the development of centralized training options and operational protocols to support installation and basic troubleshooting in-country.
- Supported the development of comprehensive service documentation, including structured service summaries and comparison tables to clarify the Pluslife offering for implementers and partners.
- Facilitated alignment on roles and responsibilities across the supply chain, encouraging a clearer definition of the responsibilities of Pluslife and its authorized distributors within country implementation models.
- Introduced the concept of diagnostic connectivity as part of the platform's longer-term integration into national health systems, highlighting opportunities to connect Pluslife instruments with existing data aggregation solutions to strengthen surveillance, diagnostic monitoring, and patient care.
- Established standardized palletization and transport parameters to improve predictability and protect product integrity.
This structured preparation reduces downstream disruption, strengthens supply continuity, and supports responsible stewardship of health resources across participating countries.
It reflects PFSCM's role not only as a supply chain operator, but as an enabler of system readiness for new diagnostic technologies.
It is systems integration in practice.
Operating at the forefront of implementation
As near point-of-care diagnostics reshape the TB response landscape, successful adoption will depend on the strength of the systems that support them.
The Pluslife introduction reflects PFSCM's role at the forefront of this shift, moving beyond transactional procurement to enable coordinated, multi-country implementation of emerging health technologies.
Ending TB will require:
- Innovation
- Market readiness
- Regulatory coherence
- Supply chain resilience
- Cross-functional execution at scale
Technology alone does not end epidemics.
"Systems do."Yes – together we can end TB.