Quality Management: Quality Assurance and Regulatory and Quality Management Systems
Commitment to quality at our coreWe take the utmost care to ensure the organizations we procure from, share our passion for-, and commitment to quality. We use a globally recognized best practice due diligence approach to quality and compliance to ensure suppliers meet the quality and regulatory standards and guidelines, such as those specified by ISO 9001, ISO 13485 and relevant European Union (EU), World Health Organization Good Manufacturing Practices (WHO GMP), and Good Distribution Practices (GDPS) requirements.
Quality Assurance and Regulatory
Our Quality Assurance & Regulatory (QA&R) unit reviews and prequalifies products for procurement which meet quality and regulatory requirements on patient safety compliance. Our QA policies cover the approval process of suppliers and products that wish to supply products.
We only procure from approved suppliers that meet the above regulatory requirements and have products that are:
Prequalified by the WHO
Approved by WHO-Listed Authority (WLA)
Recognized by founding members of the Global Harmonization Task Force (GHTF)
Compliant with applicable national regulatory requirements
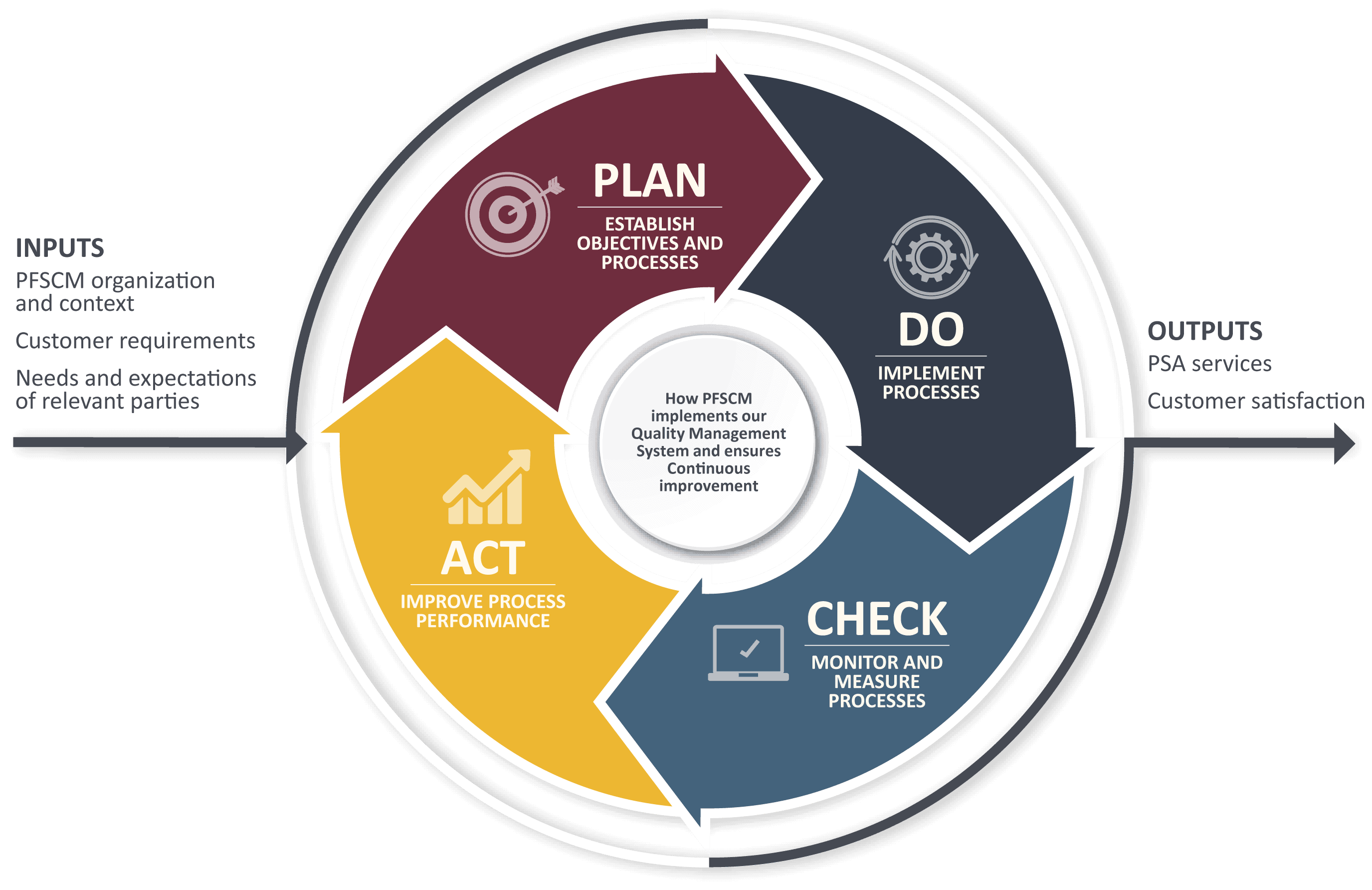
PFSCM upholds the key global principles of a QMS
Customer Satisfaction
Customer Satisfaction
A fundamental principle of ISO 9001:2015 is to continuously improve customer satisfaction by meeting and exceeding customer expectations.
Process Integration
Process Integration
The ISO process approach helps organizations to identify areas where efficiency and cost savings can be improved.
Evidence-based Decisions
Evidence-based Decisions
Through ISO 9001:2015, the organization commits to making decisions based on accurately analyzed data and facts.
Continuous Improvement
Continuous Improvement
ISO 9001:2015 requires an organization to continuously improve the adequacy, and effectiveness of its QMS performance.
Quality Management System
We are certified to ISO 9001:2015 Quality Management System (QMS). We first received our ISO 9001:2015 certification in November 2014, and have been recertified every three years since then. The certifications were awarded by one of the world’s most reputable certification bodies for management systems, DQS Management Systems Solutions. Our QMS is compliant with the WHO-MQAS and our Woerden facility holds a Wholesale Distribution Authorisation (WDA), we also are compliant with ISO 20400:2017 requirements for Sustainable Procurement.
Non-Pharma Products Prequalification Guide for Suppliers
PFSCM prequalification of non-pharmaceutical products is coordinated through our =QA&R unit. This quality and regulatory assessment of non-pharmaceutical products – through a standardized procedure is in compliance with the WHO model quality assurance system (MQAS) for procurement agencies – is aimed at determining whether a product meets our prequalification requirements and can be procured.
- The full prequalification assessment process includes the review of a full product dossier according to the quality and regulatory requirements defined for each product risk class.
- The duration of the validity of the prequalification status of a product is dependent on the manufacturer’s continuous fulfillment in meeting our quality and regulatory requirements.
- The findings are used to assess the regulatory and quality compliance of a product commercially available for procurement on behalf of our clients.
Vendors are encouraged to familiarize themselves with the prequalification steps explained in this document.
Reporting product quality incidents
Learn more
PFSCM only sources products that meet stringent, globally recognized quality benchmarks. This includes compliance with the specific quality requirements outlined in various applicable QA policies.
To uphold these standards, we collaborate with a vast network of PFSCM-approved suppliers, all of whom meet critical quality requirements, including current Good Manufacturing Practices (cGMP), ISO 13485:2016 standards for medical devices, and Good Storage and Distribution Practices. These frameworks ensure that the products we procure are manufactured, stored, and distributed under the highest quality and regulatory requirements.
PFSCM takes any deviation from product quality standards seriously. We possess the necessary expertise, systematic approach, and documented procedures to thoroughly investigate and resolve any quality issues, including out-of-specification products.
Our team is dedicated to identifying the root cause of the issue, implementing corrective actions, and preventing recurrence to maintain the integrity of our product supply chain.
In the rare event of a product quality issue, our team is ready to assist in resolving the matter efficiently. Should you encounter a product that does not meet quality specifications, we encourage you to promptly log a Product Quality Incident using form #F-QA&R-005-001-12.
You can report this by emailing productquality@pfscm.org.
Our team will provide guidance and ensure a resolution is reached through the resolution process and also ensure the situation is addressed correctly and rectified swiftly.
Download the form here: #F-QA&R-005-001-12
Contact us
Contact us to learn how we can co-create supply solutions that ensure fair access to quality health products for all.
Please send an email to sourcing@pfscm.org if you are interested in becoming a PFSCM supplier
Related
Sign up to receive public health supply chain news from PFSCM


All content ©2026 Partnership for Supply Chain Management, Inc. All rights reserved.