Who we are
PFSCM ensures lifesaving health products are accessible and available to communities in hard-to-reach places. We help donors, governments, non-profit organizations, and humanitarian agencies achieve their public health goals through end-to-end supply chain services, including cost-saving procurement, efficient logistics, pharmaceutical-grade storage, and real-time shipment tracking. PFSCM demonstrates its commitment to customer satisfaction and service excellence through our ISO 9001:2015-certified Quality Management System, which underpins all our processes, systems, and services.
PFSCM’s impact since 2007:
BILLION US$ in health products PROCURED
shipments DELIVERED, among which hundreds of cold chain products
low- and middle-income countries SERVED around the world
Latest News
Recent Blog Posts
LinkedIn Posts
What we do

Procurement services
- Best quality and value products
- Pooled procurement opportunities
- International best practices
- Navigate regulatory environment

Logistics & 4PL services
- On-time and in-full deliveries
- Cost-effective transport
- Custom and waiver clearance
- Safe, reliable shipments
Integrated supply chain services
- Tailored, real-time information
- End-to-end visibility and tracking
- Data-driven decisions
- Improved chain of custody
Recent vacancy
Accounting Lead
The Accounting Lead is responsible for providing direction and support to the accounting unit processes. The Accounting Lead is expected to manage the monthly close process including journal entries, reviews, and reconciliations of general ledger accounts.
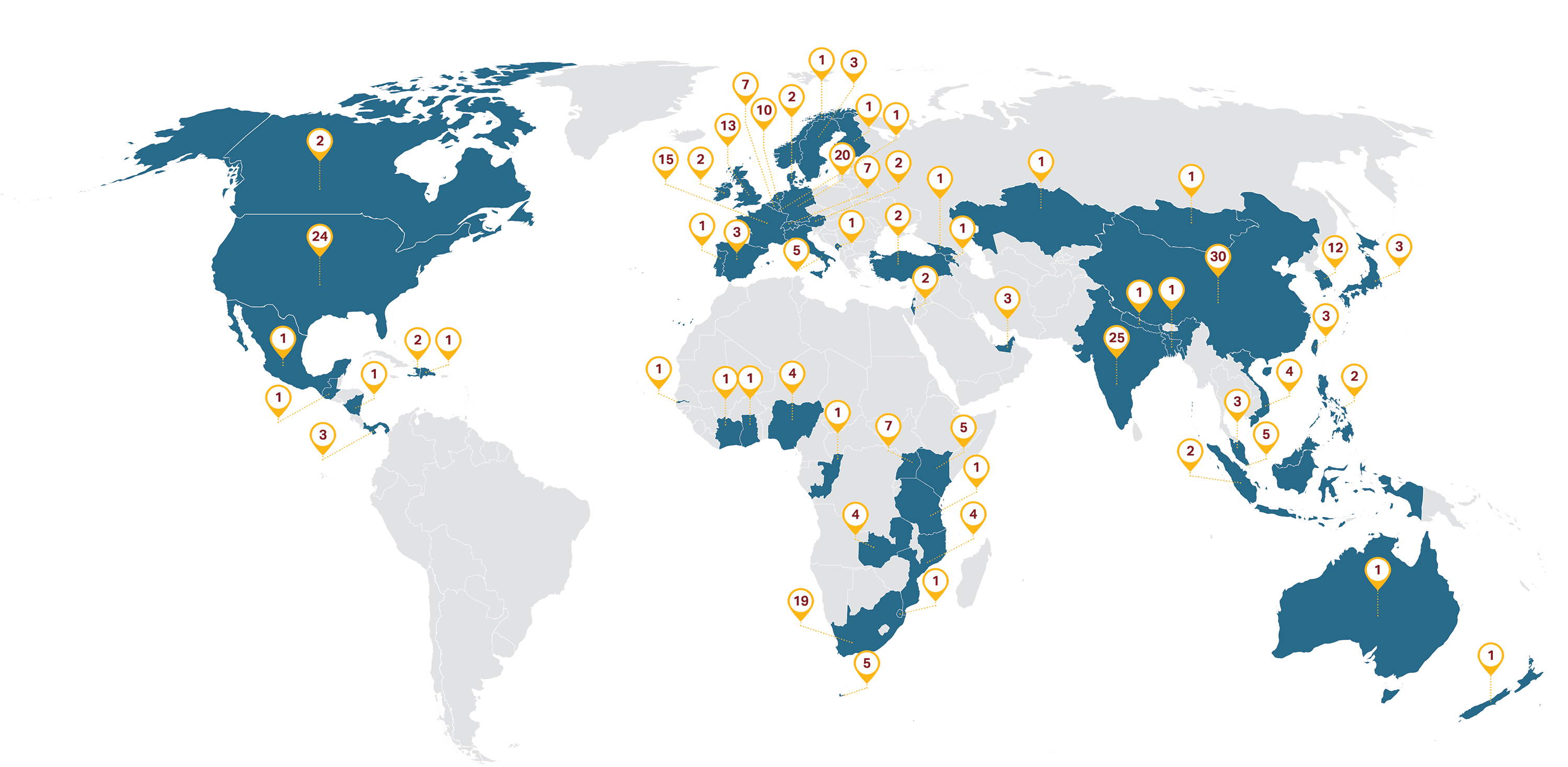
PFSCM serves clients, governments and health programs in over 100 countries, and we have access to over 290 reputable suppliers around the world.
Sign up to receive public health supply chain news from PFSCM


All content ©2026 Partnership for Supply Chain Management, Inc. All rights reserved.