Bringing faster TB diagnosis closer to patients
Each year, tuberculosis (TB) continues to infect millions of people worldwide. Yet one of the biggest challenges in controlling the disease is not treatment — it is timely diagnosis.
Globally, millions of people with TB are never diagnosed or reported to national health systems each year. These patients, often referred to as the "missing millions", may face barriers such as limited access to diagnostic services, long travel distances to laboratories, or delays in testing and reporting.
When TB goes undiagnosed, transmission continues, disease progresses, and health systems lose valuable opportunities for early intervention.
Reducing these diagnostic delays has become a central priority in the global TB response.
Recent World Health Organization (WHO) guidance supporting decentralized molecular testing and alternative sampling approaches, such as tongue swabs, reflects a growing recognition that diagnosis must move closer to where patients seek care.
Expanding access to diagnostics requires more than new technology. It requires systems capable of delivering, integrating, and sustaining those innovations across health systems.
The evolution of TB diagnostics
For decades, TB diagnosis in many low-resource settings relied primarily on sputum smear microscopy. This method is relatively simple and affordable and can be performed in peripheral laboratories and district hospitals. However, microscopy requires relatively high levels of bacteria in sputum and can miss a proportion of TB cases, particularly among children, people living with HIV, and patients with early or low-bacterial-load disease.[1]
The introduction of molecular diagnostics marked an important expansion of diagnostic capabilities. These tests can detect TB bacteria with greater sensitivity and identify certain forms of drug resistance.[2] They require stable electricity, controlled operating environments, and routine maintenance. These platforms are typically deployed in district-level laboratories or centralized diagnostic facilities, where they serve as key components of national TB diagnostic networks.
While these systems have significantly strengthened TB detection, patients in rural or underserved areas may still face delays when samples must be transported to centralized laboratories and results returned to clinics.
Emerging near point-of-care molecular diagnostic platforms represent another step in strengthening the diagnostic network. Designed to operate in decentralized environments, these smaller and more flexible systems can support molecular testing closer to primary care facilities, district hospitals, and community-level health services.
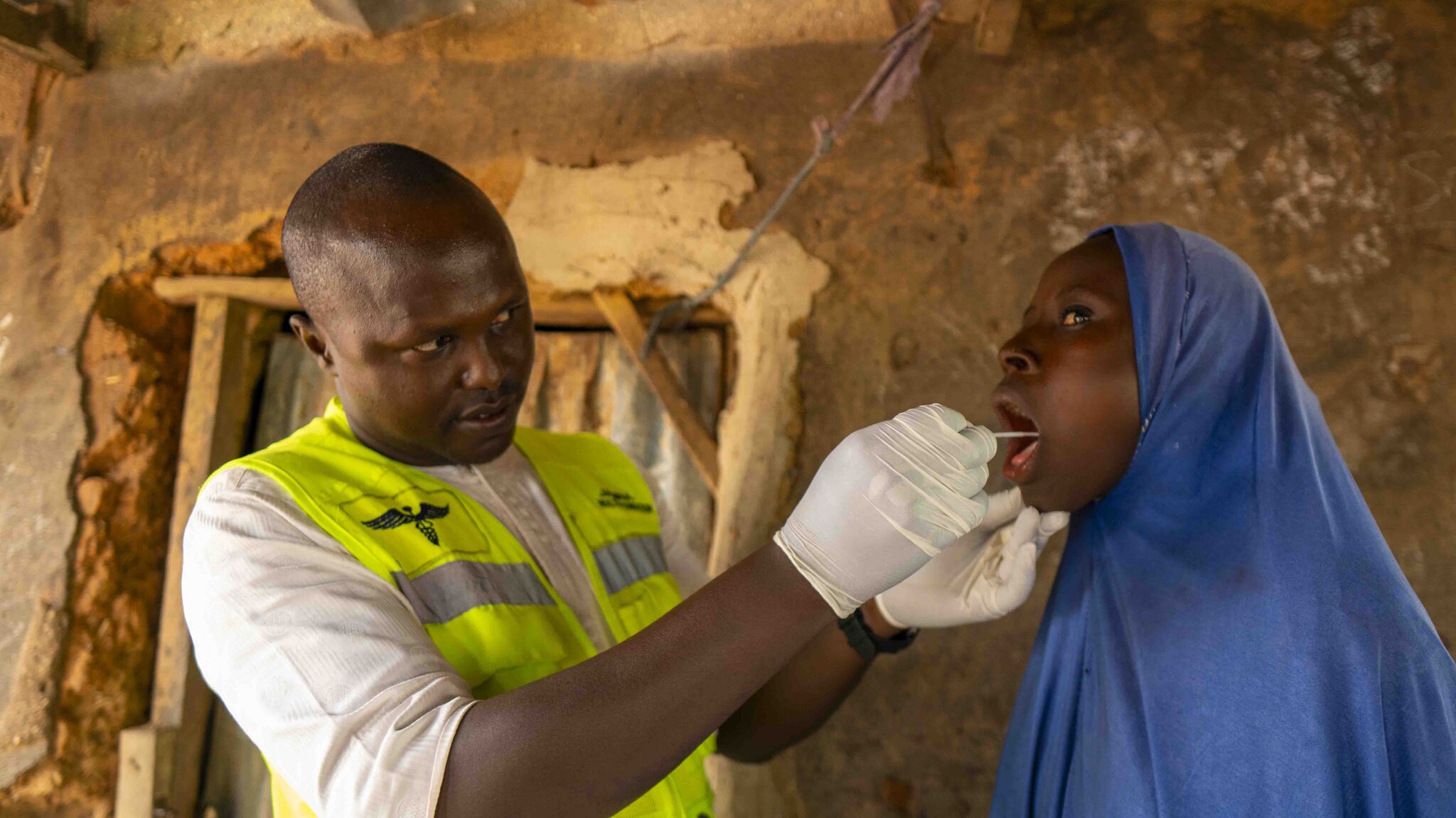
When paired with innovations such as tongue swab sampling, these platforms may also expand testing access for populations that have historically been more difficult to diagnose — particularly children,[3] who often cannot produce sputum samples required for traditional testing approaches.
Rather than replacing existing technologies, these newer platforms expand the range of tools available to TB programs. By supporting molecular testing at different levels of the health system — from centralized laboratories to decentralized care settings — they help strengthen diagnostic networks and bring faster TB detection closer to the communities most affected by the disease.
This is where supply chain and implementation systems become critical.
Preparing the systems behind the technology
Across 13 countries participating in early implementation efforts, the Partnership for Supply Chain Management (PFSCM) is supporting preparations for the introduction of the Pluslife MiniDock Ultra, a near point-of-care molecular TB diagnostic platform capable of delivering results in about 30 minutes.
Designed for decentralized environments, the platform can operate in settings with high temperatures and humidity and supports both sputum and tongue swab samples.
The inclusion of tongue swab sampling reflects evolving diagnostic guidance and could significantly expand access to testing for children and other patients who cannot easily produce sputum samples.
Diagnosing tuberculosis in children has long been one of the most difficult challenges in TB control. Young children often cannot produce sputum samples required for conventional diagnostic tests, and the disease frequently presents with lower bacterial loads, making it harder to detect using traditional methods. As a result, paediatric TB is widely considered underdiagnosed and underreported.[4] Alternative sampling approaches such as tongue swabs may help address this challenge by enabling easier and less invasive sample collection in primary care settings.[3] Expanding diagnostic options for children is an important step toward closing persistent gaps in TB detection and ensuring that young patients are diagnosed and treated earlier.
For national TB programs, improving paediatric diagnosis is essential to closing the global TB detection gap.
However, introducing new diagnostic technologies across multiple countries requires significant preparation before the first shipment ever moves.
PFSCM has supported this preparation through cross-functional coordination across logistics, sourcing, quality assurance, and client services, alongside collaboration with the Global Fund.
Teams have also worked through regulatory questions related to evolving WHO TB diagnostic guidance, helping ensure alignment between global policy recommendations and country-level implementation plans.
Resolving these issues early helps prevent regulatory bottlenecks that can delay the introduction of new technologies into countries.
Developing supply chains for diagnostic access
In parallel with regulatory preparation, PFSCM's logistics team has been preparing and coordinating the end-to-end shipment processes needed to support country orders — from supplier pickup through delivery.
A key focus has been load optimization and considerations for shipment planning — technical work that ensures the Pluslife equipment can move efficiently across international supply chains.
Redesigning pallet configurations and aligning shipments with international load standards, PFSCM teams increased pallet capacity from 500 kilograms to 1,500 kilograms, enabling significantly more equipment to be transported safely within each shipment.
These changes translate into real operational gains.
Improved load optimization allows:
- More diagnostic devices per shipment
- Fewer shipments are required overall
- Lower international transport costs
- Reduced risk of product damage
- More efficient use of donor-funded resources
Even before country orders are finalized, teams have developed draft transport plans and mapped country-specific import documentation requirements to reduce potential delays once shipments begin.
These behind-the-scenes preparations may not be visible to patients, but they directly influence how quickly diagnostic technologies reach the facilities that need them.
Reducing costs while expanding diagnostic reach
Decentralized diagnostic technologies also have important cost implications for health systems.
Traditional molecular testing platforms require centralized laboratories, dedicated infrastructure, and specialized maintenance. These systems are highly effective but can be expensive to deploy at scale across remote or rural areas.
Near point-of-care platforms can help expand diagnostic capacity at lower levels of the health system, reducing the need for extensive sample transport networks and shortening diagnostic timelines.
Earlier diagnosis can also reduce the long-term costs of TB care.[5]
When TB is detected earlier:
- patients begin treatment sooner
- transmission in communities is reduced
- severe or advanced disease is prevented
These improvements can help reduce both health system costs and patient burdens, while strengthening national TB control programs.
These behind-the-scenes preparations may not be visible to patients, but they directly influence how quickly diagnostic technologies reach the facilities that need them.
Expertise makes new innovations work
Technological innovation has transformed the global TB response over the past two decades. However, new diagnostic tools alone cannot end an epidemic.
As preparations move toward implementation, the coming months will provide important opportunities to learn how decentralized diagnostic approaches can support national TB programs.
PFSCM looks forward to continuing to support these efforts and to seeing how these developments contribute to the broader global effort to improve TB diagnosis and reach the communities most affected by the disease.
References
- WHO. Xpert MTB/RIF implementation manual. https://www.who.int/publications/i/item/9789241506335
- WHO. Xpert MTB/RIF implementation manual (drug resistance detection). https://www.who.int/publications/i/item/9789241506335
- Wood RC et al. Detection of Mycobacterium tuberculosis in tongue swabs. Clinical Infectious Diseases. https://academic.oup.com/cid/advance-article/doi/10.1093/cid/ciag077/8501003
- WHO. Global Tuberculosis Report 2025. https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports/global-tuberculosis-report-2025
- WHO. TB Reports. https://www.who.int/teams/global-tuberculosis-programme/tb-reports