The Partnership for Supply Chain Management (PFSCM) through its partner Celsian Consulting and parent company John Snow Inc., recently initiated a Center of Excellence (CoE) program to assist the Ethiopian Pharmaceuticals Supply Agency (EPSA) in implementing private sector practices to improve its operational performance and maturity in preparation for future ISO certification.
EPSA, a government organization established 70 years ago, manages a wide network of clusters that comprise a mix of warehouses and office locations throughout Ethiopia. Through these clusters, it supplies pharmaceuticals and medical equipment mainly to government health institutions.
EPSA has recently undergone a transformation to become more efficient and data-driven, and is working to expand and mature its capabilities to eventually achieve quality management certifications such as ISO 9001:2015 and others.
Celsian Consulting, a supply chain consultancy firm specializing in adapting private sector solutions for public health supply chains, worked in country with EPSA for almost three years to establish a CoE that will help the agency achieve its goals in a sustainable manner using existing infrastructure, personnel and systems.
Celsian Consulting’s mission was to help EPSA implement private sector practices and standards to achieve higher levels of performance without EPSA having to invest heavily in technology or human resources.
Background and scope
The CoE program started in 2018 with a proof of concept, which focused on EPSA’s primary hub in Adama, and its four Central Medical Stores that surround this hub in the area extending up to Addis Ababa.
Following the successful proof of concept phase, the full program, which was funded by PFSCM through a major global donor, was kicked off in September 2019, and it ran until July 2021.
For the full program, support was provided at the primary hub in Adama, the four Central Medical Stores, and eight other auxiliary warehouses which were set up to accommodate overflow.
In addition, seven other hubs, one each in Bahir Dar, Dessie, Dire Dawa, Hawassa, Jimma, Mekelle, and Nekemte were also included in the full program.
|
EPSA warehouses |
|
| Tier 1 |
|
| Tier 2 |
|
| Tier 3 |
|
The full CoE program started with comprehensive baseline assessment of each of the warehouses. A dedicated team was based at EPSA’s Central Medical Stores from September 2019 to March 2021, a total of 18 months.
Between September 2019 and December 2019 the team:
- developed the baselines of each Central Medical Store and primary hub,
- established individual implementation plans,
- established the Key Performance Indicators that would be measured,
- and created the reporting templates and CoE governance structures.
The team rigorously tracked the performance achieved by each EPSA warehouse between January 2020 to March 2021.
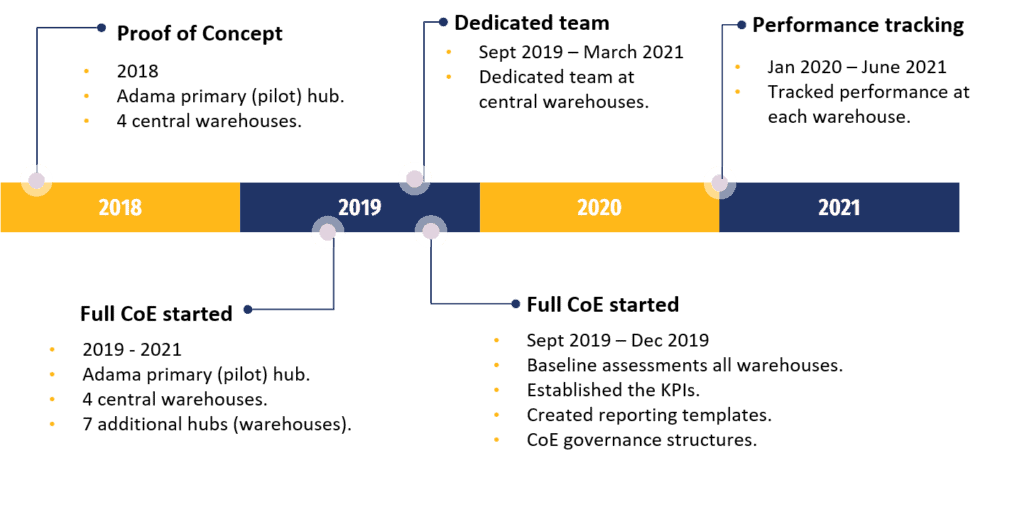
Figure 1: CoE Timeline
The challenges faced
Overall, from the assessments done, it was determined that EPSA struggled with master data collection and management; and had limited visibility into inventory levels across its various warehouses. The shortcomings in data- and inventory management, and insufficient adherence to good storage and distribution practices resulted in several supply chain issues such as: low stock availability, long delivery schedules, a high number of annual stock counting days and expired stock.
EPSA’s objectives
EPSA’s vision is to become Africa’s leading public health supply chain organization. To achieve this, EPSA must become data- and performance-driven, and must improve its operational maturity to prepare for ISO 9001:2015 certification.
The CoE approach and solutions
The CoE approach entailed the use of Celsian Consulting’s GRIDD methodology that improves supply chains in a sustainable, independent way.
GRIDD has been strengthened over 15 years by distilling experience from over twenty countries into a robust methodology that is scalable and effective in measuring and improving supply chain networks’ levels of maturity.
For EPSA, the methodology involved:
- Deploying a custom-built maturity modeling toolkit for each hub in a “phased gate” manner that requires each project stage to be reviewed before moving on to the next.
- Using Lean Sigma Six principles to improve performance by systematically removing waste and reducing variation through collaborative team effort.
- Developing KPIs, which are critical measurements, needed to inform and track change and improvements.
|
Maturity Level |
Maturity Level Outcome |
|
Level One |
Developing supply chain functionality. |
|
Level Two |
Basic operational effectiveness. |
|
Level Three |
Organizational functionality and good governance structures. |
|
Level Four |
Management operating systems and LEAN operational performance. |
|
Level Five |
Quality Management System (ISO 9001:2015 audit-ready) and optimal business economics. |
In terms of KPIs, the CoE program developed two overarching sets of measurements:
- operational performance and,
- compliance with Good Storage Practices (GSP) and Good Distribution Practices (GDP) as set out by the World Health Organization.
Operational performance was tracked through three sub-KPIs namely:
- stock availability in the system that can be attributed to quick receipt and put away,
- inventory accuracy, and
- conformance to the distribution plan.

Before and after photos of cleaning at one of the primary hubs in Adama
CoE outcomes and achievements
Starting from the proof of concept period, the CoE program has delivered significant and measurable improvements.
Overall, the CoE improved EPSA’s visibility into their operations, in turn enabling them to make better data-driven decisions to improve their operational performance.
Improvements were achieved in four key areas:
-
-
- Data management
- Inventory management
- Warehouse management
- Maturity growth
-
Data management
Accurate, usable data and information is a key supply chain driver and is crucial to EPSA’s current and future operations. Accurate data supports the cohesive operation of all other supply chain activities, such as facility- and inventory management, distribution, and procurement.
Through the CoE, EPSA has:
- Improved master data management at the product level by collecting product profile information and volumetrics for its 1,600 active SKUs.
- Developed information collection tools for key warehouse activities associated with the receiving of stock into EPSA’s network, and the further distribution of stock into the regions.
- Developed a Personnel Development Plan.
Meanwhile, the majority of the program execution took place during the outbreak of COVID-19. The CoE was well positioned to develop solutions to overcome COVID-19 specific supply chain issues.
Several warehouse operations reports were customized to provide EPSA’s COVID-19 Task Force team with real-time network storage capacity information. This vital information allowed the COVID-19 Task Force team to quickly make informed decisions, improve distribution planning, and schedule inbound COVID-19 products while they were still in transit to Ethiopia.
Late in 2020, at the height of the COVID-19 pandemic, the team supported EPSA to measure open pallet locations within each warehouse to provide an accurate view of overall network capacity.
This measurement continues to be helpful to EPSA, both for regular pharmaceuticals and medical supplies and for the government’s COVID-19 response.
Inventory management
Reliable inventory management is an essential contributor to enhancing supply chain performance. The CoE implemented several private sector best practices to develop capacity, improve the accuracy and efficiency of inventory management, and ensure accurate put-away, and the efficient use of warehouse space.
|
Impact Figures |
Area of accomplishment |
Description |
| 50% reduction | Delivery lead time | The delivery schedule between the Adama hub and the health facilities that it supports was reduced from eight weeks to four weeks (the pilot program). Improving last-mile delivery was the ultimate realization of upstream improvements. |
| 68.5% improvement | Lead time for dock-to-stock
(The process that begins when the warehouse receives goods at the loading docks and ends once they have been stored on the racks) |
Lead times for dock-to-stock were tracked weekly and reduced from 12 days to 3.8 days over ten weeks. This daily measurement formed a vital part of the overall maturity growth of EPSA.
|
| 49% improvement | Inventory Accuracy | At the Central Medical Stores inventory accuracy improved from 38% to 74%. This improvement was achieved over nine months and included maintaining inventory accuracy above 95% for the last five months of the program. |
| 23% improvement | Inventory Accuracy | At the primary hubs inventory accuracy improved from 80% to 98% This improvement was achieved over eight months, with the maintenance of inventory accuracy above 95% for the last five months. |
| 88% improvement | Annual stock counting days | Annual stock counting days were reduced from 42 days to 5 days. |

Preparing for the annual stock count at one of the primary hubs
Warehouse management
At the start of the program, EPSA’s Central Medical Stores included a combination of eight auxiliary warehouses and eight more off-site warehouses. Managing and keeping track of stock across these sites and warehouses was a significant challenge, and it affected overall operations, particularly for the visibility of accurate stock levels at each location.
The CoE program earmarked warehouse optimization as a critical area, and the results of the interventions had a notable impact across the supply chain. To improve the operational performance and maintain maturity growth, the CoE reduced the complexity and costs associated with the auxiliary warehouses.
| Interventions |
Outcomes |
| Fast-Normal-Slow analysis of the program and revolving drug fund stocks.
|
|
| ABC analysis and operationalizing new warehouses. |
|
| Reviewed and updated expired stock and disposal processes. |
|
As a result of these activities, six of eight auxiliary warehouses were closed, and the stock from these warehouses was integrated into the Central Medical Stores.
At the Central Medical Stores, the GSP and GDP improved by 20% year on year, from 52% in February 2020 to 72% in February 2021, and even more to 80% in March 2021.
Maturity growth
When the CoE pilot was done in 2018, only two of the 12 warehouses (Adama, four Central Medical Stores and seven primary hubs) were at Maturity Level Two.
By February 2021, seven warehouses had progressed to Maturity Level Two, and three excelled to Maturity Level Three.
When Celsian Consulting handed the CoE over to EPSA to operate independently in July 2021, all warehouses were on track to reach Maturity Level Three. Since then, EPSA continued to implement the CoE approach and GRIDD methodology at its other warehouses, and recently one of the secondary warehouses reached Maturity Level Five and earned its ISO 9001:2015 certification.
With continued funding and support, EPSA can continue along this trajectory to achieve Maturity Level Five across all its warehouses, making their quality management system audit ready for ISO 9001:2015 certification.

Figure 2: Number of warehouses that progressed to Maturity level Two and Three
Overall, by February 2021, EPSA’s maturity growth improved on average 12.5%.
This key measure can be further apportioned:
- Four Central Medical Stores at 13.0%
- Seven primary hubs at 11.2%

Figure 3: Maturity growth measured at warehouses
Key takeaways
The CoE approach has successfully demonstrated that EPSA’s existing infrastructure and human resources can be optimized to achieve higher output levels to improve product availability, while minimizing service costs and product wastage.
Further, implementing private sector practices through a proven methodology delivers measurable results, but funding, donor buy-in, and political will is needed to identify and create such partnerships.
In addition, the GRIDD methodology has proven to transform supply chain networks into sustainable systems that can operate at first world, best practice levels.
Meanwhile, EPSA’s maturity progress is highly encouraging, and the ISO 9001:2015 certification of one of the secondary hubs is testament to the effectiveness of the CoE. Assuming the current maturity growth momentum is maintained, four of the primary hubs can also be expected to pursue ISO 9001:2015 accreditation soon.
Conclusion
The CoE program can deliver long-term sustainability with financial benefits to the Ministry of Health and donors.
For example, by increasing the annual stock turn rate from 0.89 to 1.5 per annum, EPSA’s storage capacity is extended by about 10 years.
This puts money back into health program budgets with minimal or no infrastructure or system investments.
Assuming funding continues, the CoE program’s contributions will continue to support improvements in EPSA, positioning it as Africa’s leading public health supply chain organization.

The photo on the left shows one of the primary hubs before intervention, and the two photos to the right show the warehouse with GSP and GDP best practices implemented.