By Tim Meehan, PFSCM Senior Laboratory Technical Advisor.
People Who Inject Drugs (PWID) are a key population of focus in stemming the transmission of viral hepatitis and other blood-borne pathogens. When products such as syringes and needles are shared between users, person-to-person transmission of pathogens can occur. According to The Lancet Global Health, in 2017, the global prevalence of Hepatitis B (HBV) among PWID was 76%, and that of Hepatitis C (HCV) was 88%. Access to injection drug use harm reduction (HR) tools, such as low-dead space needles and syringes, is an important part of the public health approach to mitigate the transmission of viral hepatitis.
The controversial nature of HR presents unique obstacles to programs, including product availability and logistics. The challenges in procuring high-quality products which meet local usage preferences can be especially problematic for nascent programs in low- and middle-income countries (LMICs).
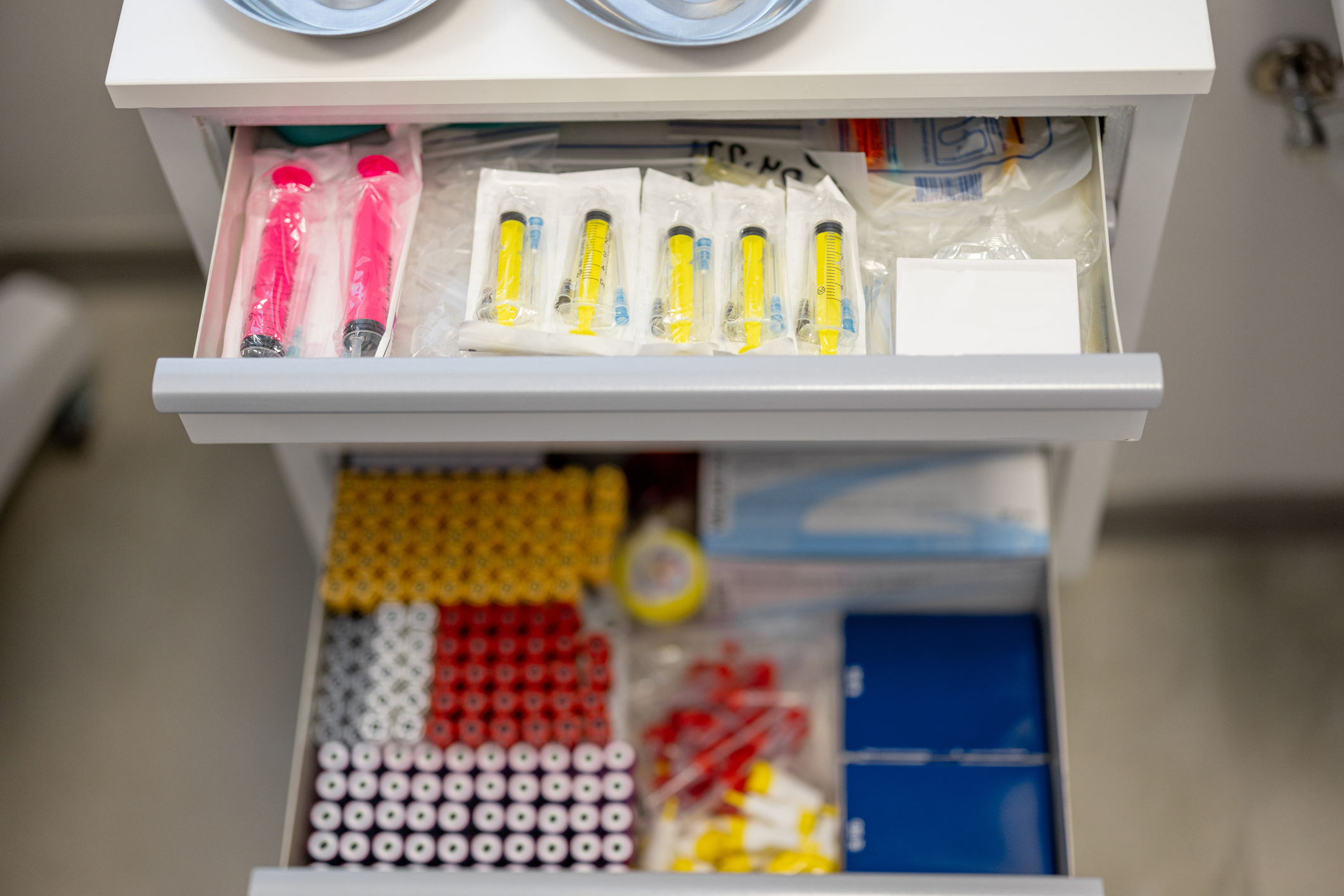
General use products, such as generic syringes, are often used when specific HR products are not available. However, generic products do not have features that specifically mitigate the transmission of blood-borne diseases. The range of products used for HR is complex and varies widely based on location. The most appropriate products for a given region are often specific to local preferences, the types of drugs being used, and law enforcement practices. The initial product categories which will soon be available for procurement from PFSCM include:
- Needles and syringes in a range of gauges, lengths, and volumes (including Low Dead Space (LDS) syringes with attached needles, LDS detachable needles, and syringes)
- Drug preparation cookers
- Acidifiers
- Water for injection
- Filters
- Tourniquets
- Disinfectant swabs
PFSCM is taking a holistic approach to expanding our portfolio of HR products. To obtain the understanding needed to provide the best products for a range of locations and scenarios, we have sought input from stakeholders across the HR spectrum, such as civil society and grassroots organizations, Ministries of Health, and international non-governmental organizations. Through these engagements, PFSCM is developing a nuanced understanding of the particular products which are necessary in specific areas.
PFSCM is also engaging directly with suppliers to ensure the best value. As a part of our supplier engagement, we apply a stringent process to assess the reliability of manufacturers and the quality of their products.
PFSCM’s collaborative engagement with all stakeholders will allow us to provide a global, comprehensive package of products which is harmonized to meet local needs, with quality-assured (QA) products to maximize the HR impact, and at a price point which is acceptable in an LMIC context.
____________________________________________________________________________________________________________________
Global prevalence of injecting drug use and sociodemographic characteristics and prevalence of HIV, HBV, and HCV in people who inject drugs: a multistage systematic review, Degenhardt et al., 2017; 5: e1192–20.
World Hepatitis Day – 28 July 2028
At PFSCM, we stand united on World Hepatitis Day to raise awareness about this preventable disease. Our commitment to supporting global health extends to ensuring a smooth and efficient flow of lifesaving products and tools in the fight against hepatitis.
Today we celebrate the relentless efforts of healthcare professionals worldwide who work tirelessly to combat hepatitis. Through collaboration and innovation, we can make a tangible impact in controlling the spread of hepatitis and improving public health.