diagnostics (2020–2025)
coordinated
procurement value
products delivered
Yes! We Can End TB: strengthening global health supply chains to expand TB diagnosis
The theme of World TB Day 2026 — "Yes! We Can End TB! Led by Countries. Powered by People." reflects a global commitment to accelerate progress toward ending tuberculosis (TB).
Many countries are working to expand TB screening programs, strengthen diagnostic networks, and improve access to treatment as part of global efforts to end the disease. Achieving these goals requires more than medical innovation — it also depends on strong and resilient global health supply chains capable of procuring, transporting, and delivering tuberculosis diagnostics and lifesaving technologies to health systems worldwide.
These TB diagnostic supply chains play a critical role in ensuring that new diagnostic technologies can be introduced, scaled, and sustained within national health systems, while existing infrastructure can also still be maintained and supported.
As a trusted global health procurement and logistics services provider, the Partnership for Supply Chain Management (PFSCM) works with governments, donors, and global health partners to strengthen TB supply chains and ensure countries have timely access to quality-assured diagnostic tools.
Through strategic procurement, coordinated logistics, and digital supply chain solutions, PFSCM helps countries expand TB detection, improve diagnostic efficiency, and maximize the impact of health investments.
Stronger diagnostic supply chains mean more screening, earlier diagnosis, and better visibility of the TB epidemic.
With countries leading the response and communities driving progress, yes — we can end TB.
PFSCM — World TB Day 2026Key takeaways
PFSCM supports countries to expand tuberculosis diagnosis through (2020–2025):
Global procurement and delivery of TB diagnostic technologies across 63 countries.
Coordinating 427 international shipments of TB diagnostic equipment, reagents and consumables.
Managing specialized logistics for time and temperature-sensitive molecular diagnostics.
Supporting digital procurement through the HealthSupply Portal.
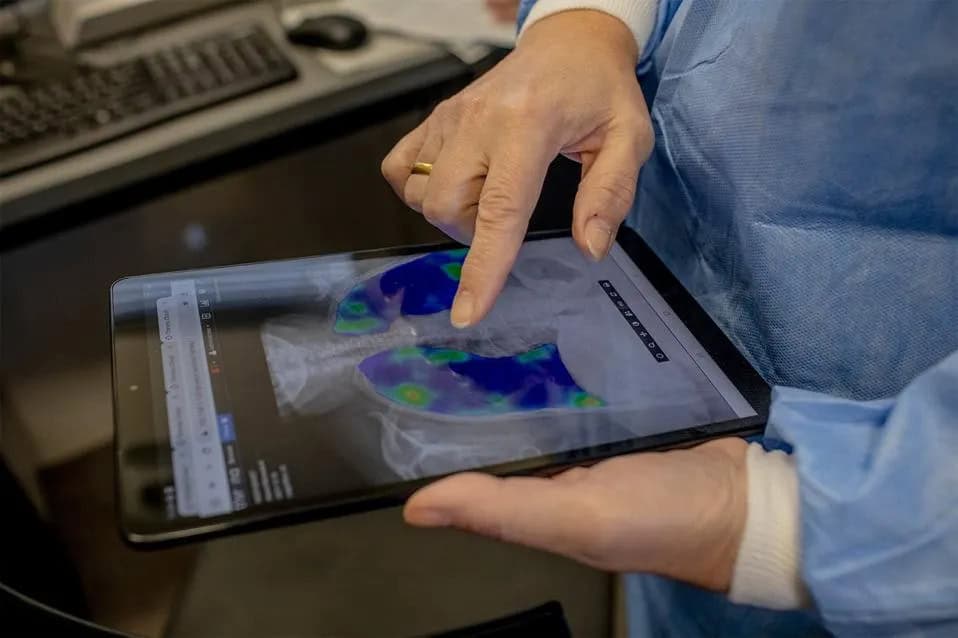
Helping countries scale innovative technologies such as computer-aided diagnosis/detection (CAD) and AI-enabled X-ray screening, and near point-of-care molecular diagnostics.
Delivering TB diagnostics at a global scale
PFSCM supports TB programs and global health partners by procuring and delivering quality-assured tuberculosis diagnostic technologies through integrated coordinated global supply chains.
Between 1 January 2020 and 31 December 2025, PFSCM delivered TB-related health products to 63 countries, coordinating 427 international shipments and delivering 270,126 units of TB diagnostic products and equipment, representing $146.4M in procurement value across PFSCM clients.
These shipments included a wide range of TB diagnostic technologies, such as:
- Molecular TB diagnostic analyzers designed to perform nucleic acid amplification tests (NAAT) and polymerase chain reaction (PCR).
- Drug-resistance testing assays for rifampicin and isoniazid detection.
- Rapid diagnostic tests (RDTs).
- Laboratory equipment, such as microscopes and consumables, and reagents.
- Fixed, mobile, portable, and ultra-portable digital X-ray screening systems.
- CAD AI software for TB screening.
Together, these technologies support national TB diagnostic networks at multiple levels of the health system — from centralized laboratories performing molecular testing to community screening programs reaching underserved populations.
For Ministries of Health and global health partners, this global delivery footprint demonstrates PFSCM's ability to manage complex TB supply chains across diverse operating environments while ensuring that diagnostic tools reach health facilities quickly and reliably.
Managing complex TB diagnostic supply chains end-to-end
Sourcing and procurement
At the start of the supply chain, PFSCM supports national TB programs through strategic sourcing, procurement, and quality assurance. This includes market analysis, supplier due diligence, and verification of compliance with international quality standards to ensure that diagnostics and related technologies are safe, effective, and fit for program needs.
Through competitive tendering, long-term framework agreements, and active supplier negotiations, PFSCM strengthens purchasing power and secures best-value pricing for global health programs. Over the past five years, these strategic sourcing efforts have generated more than $5M in cost savings for medical imaging equipment and associated computer-aided detection (CAD/AI) technologies, helping countries expand access to TB screening while making efficient use of donor funding.
Logistics
TB-related products can involve highly specialized logistics.
Products range from large digital imaging systems to time- and temperature-sensitive molecular reagents, each requiring specific transport and storage conditions.
These include molecular diagnostic reagents, cartridges, and controls used in TB testing platforms (such as NAAT and PCR-based systems), which have specific handling and stability requirements.
Many molecular diagnostic reagents require cold or frozen storage to maintain stability. Some components must be kept at –20 °C, while others require refrigerated conditions (2 °C to 8 °C). In certain cases, particularly for long-term storage of sensitive materials, temperatures as low as –80 °C may be required. These requirements make temperature-controlled logistics critical to preserving reagent integrity during international transport and storage.
Maintaining these conditions is critical to ensuring test accuracy, quality assurance, and reliable diagnostic results, as temperature excursions can compromise performance.
PFSCM manages these complexities through specialized health logistics planning, including:
- temperature-controlled shipping solutions
- route planning based on airport and warehouse cold-chain infrastructure
- temperature monitoring and data loggers
- proactive customs and clearance coordination
PFSCM supports this work through a global logistics network spanning more than 3,000 established shipping lanes, working with specialized third-party logistics providers (3PLs) experienced in global health logistics selected for their expertise in specific regions, transport modalities, and cold-chain requirements.
Pre-negotiated transport agreements help reduce transit risks, improve cost predictability, and ensure sensitive diagnostics move through international supply chains with greater reliability.
These safeguards help protect high-value diagnostics, reduce product loss, and ensure national TB programs can maintain uninterrupted testing capacity.
Turning procurement efficiency into expanded screening
Through strategic sourcing and best procurement practices, including leveraging framework agreements with key manufacturers, PFSCM helps drive down the cost of TB diagnostic products and related services, thereby expanding access through improved affordability.
An analysis conducted by PFSCM on digital X-ray systems and CAD/AI software procured between 2021 and 2025 showed cost savings of more than $5.4M. These savings have directly contributed to expanding diagnostic services in low- and middle-income countries (LMICs).
The savings generated could enable the deployment of approximately 82 additional portable digital X-ray systems with CAD/AI, helping national TB programs expand screening coverage. Based on operational assumptions of the number of scans a system can perform per day, these additional systems could support more than 2.5 million additional TB screening scans per year in case-finding efforts in affected countries. Earlier detection and diagnosis through expanded screening can help limit the spread of TB and improve treatment outcomes.
Increasing screening coverage, particularly in rural and underserved communities where access to diagnostic services is limited, supports global efforts toward TB elimination goals. Expanded screening also improves visibility of the TB disease burden, enabling countries to identify previously undiagnosed cases and better target interventions.
These savings have directly contributed to expanding diagnostic services in low- and middle-income countries (LMICs).
Equivalent to approximately 82 additional portable digital X-ray systems, supporting more than 2.5 million additional TB screening scans per year.
Expanding TB screening capacity in Nigeria
Large-scale diagnostic deployments illustrate how coordinated procurement, global logistics, and in-country distribution planning can strengthen national TB detection programs.
Expanding TB screening capacity in Nigeria
In Nigeria, PFSCM supported the procurement and delivery of TB diagnostic equipment and services valued at more than $37M, coordinating 29 international shipments between 2024 and 2025. This enabled the deployment of molecular TB diagnostic technologies across 333 sites, alongside the nationwide rollout of 370 digital X-ray systems.
The deployment included:
- 370 portable digital X-ray systems and AI-enabled diagnostic software
- molecular diagnostic platforms capable of rapid tuberculosis detection and drug-resistance testing, including Truenat systems
These portable imaging systems support large-scale TB screening, while molecular diagnostic platforms enable rapid bacteriological confirmation and drug-resistance detection within the diagnostic pathway.
By early 2026, 370 digital X-ray systems had already been delivered and installed, significantly expanding TB screening capacity across multiple regions.
PFSCM's role extended beyond procurement and international transport. Working with a dedicated in-country Nigeria team, PFSCM supported importation and regulatory processes, including coordination with national authorities such as NAFDAC, SONCAP, and IDEC, as well as the management of customs waivers and documentation. The project also involved coordinating installation across hundreds of sites, equipment warranties, maintenance planning, training for local health workers, and ongoing software and service renewals to ensure the systems remain operational over time.
Expanding access to diagnostic technologies through reliable supply chains helps national programs detect tuberculosis earlier and reach more communities with lifesaving screening services.
Ensuring innovation in TB diagnostics
Advances in diagnostic technology are transforming how countries detect tuberculosis.
Portable and ultra-portable digital X-ray systems, combined with CAD artificial intelligence, are helping countries scale community-based screening programs and accelerate the interpretation of chest X-rays.
At the same time, near point-of-care molecular diagnostics are bringing high-sensitivity TB testing closer to patients.
Portable molecular platforms allow testing to be conducted in decentralized facilities and community screening sites, reducing the time between testing, diagnosis, and treatment initiation.
PFSCM supports the introduction and scale-up of these innovations by ensuring new technologies can be procured, transported, and deployed effectively within national health systems.
One example, from a new portfolio PFSCM has been developing is, Pluslife, a new near point-of-care molecular diagnostic platform designed to expand access to rapid TB detection in decentralized settings.
Preparing for the rollout of technologies like Pluslife requires extensive supply chain planning — from supplier onboarding and packaging design to freight strategies, customs documentation, and in-country distribution planning.
By aligning procurement, logistics planning, and implementation timelines early, PFSCM helps ensure innovative diagnostics move efficiently from production to the health facilities where they can deliver the greatest impact.
Recent WHO recommendations supporting decentralized TB testing and near-point-of-care diagnostics are accelerating the need for supply chains capable of rapidly and reliably delivering new diagnostic technologies.
Strengthening procurement through PFSCM's HealthSupply Portal
Efficient procurement systems are essential for ensuring countries can quickly access the diagnostic technologies they need.
PFSCM also supports procurement efficiency through digital tools such as the HealthSupply Portal, PFSCM's online procurement platform designed specifically for global health supply chains.
The portal provides streamlined access to more than 6,200 quality-assured health products, allowing countries and implementing partners to search, quote, order, and track shipments through a secure digital interface connected to PFSCM's global supply chain control tower.
By improving procurement visibility, product selection, and order management, the platform helps countries reduce procurement lead times and strengthen supply chain transparency.
Supplier access programs support regional and global diagnostics sourcing across multiple disease and health areas, including TB, HIV, human papillomavirus, viral hepatitis, genomic surveillance, and antimicrobial resistance (AMR).
These digital procurement tools improve visibility across supply chains while helping countries access quality-approved TB products more efficiently.
quality-assured health products available through the HealthSupply Portal
PFSCM's HealthSupply Portal
PFSCM's online procurement platform designed specifically for global health supply chains — allowing countries and implementing partners to search, quote, order, and track shipments through a secure digital interface connected to PFSCM's global supply chain control tower.
Visit HealthSupply Portal →Supporting TB diagnosis across the diagnostic pathway
Effective TB detection depends on integrated diagnostic networks operating across multiple levels of the health system. PFSCM's TB product portfolio supports this continuum by providing countries with the diagnostic tools needed from community screening to centralized laboratory testing.
Screening & Triage
At the screening and triage stage, typically conducted at community and primary care levels, portable technologies such as AI-enabled digital chest X-ray systems support outreach and active case-finding by identifying individuals who may require further diagnostic testing.
Bacteriological Confirmation
At the bacteriological confirmation stage, molecular diagnostic platforms enable rapid, high-sensitivity detection of TB infection. These platforms — often deployed at district or regional facilities — include technologies based on nucleic acid amplification testing (NAAT) and polymerase chain reaction (PCR), which allow TB programs to confirm infection and initiate treatment more quickly.
Drug-Resistance Testing
For drug-resistance testing and advanced laboratory analysis, central and reference laboratories use additional diagnostic methods, including molecular line probe assays, culture, and phenotypic drug susceptibility testing to determine whether patients require first-line or second-line treatment regimens.
By supporting the procurement of diagnostic technologies, reagents, and consumables across these different stages of the diagnostic pathway, PFSCM helps countries strengthen integrated TB diagnostic networks and improve the pathway from screening to diagnosis and treatment.
Detecting and managing drug-resistant TB
Drug-resistant tuberculosis presents one of the most significant challenges in global TB control. Identifying resistance to key medicines such as rifampicin and isoniazid is critical for determining appropriate treatment regimens and preventing the spread of resistant strains.
PFSCM supports access to diagnostic technologies capable of detecting drug-resistant TB, including rapid molecular tests that identify rifampicin resistance, as well as molecular line probe assays and culture-based drug susceptibility testing used in reference laboratories.
Ensuring access to these diagnostics allows national TB programs to detect drug resistance earlier and guide appropriate treatment decisions. Strengthening access to drug susceptibility testing improves patient outcomes, supports more effective treatment strategies, and helps protect the long-term effectiveness of existing TB medicines.
Strengthening supply chains to End TB
Ending tuberculosis requires more than new technologies — it requires health supply chains capable of delivering those technologies reliably and at scale.
Through cost-effective and pooled procurement, digital procurement tools, technical product expertise, and specialized health logistics, PFSCM helps countries expand access to TB diagnostics while ensuring efficient use of global health resources.
As the global community marks World TB Day 2026, the message is clear:
Stronger diagnostic supply chains mean more screening, earlier diagnosis, and better visibility of the TB epidemic.
With countries leading the response and communities driving progress, yes — we can end TB.
"Yes! We Can End TB."